Corrective and Preventive Action (CAPA) Procedure for GMP
GMPSOP
MARCH 17, 2023
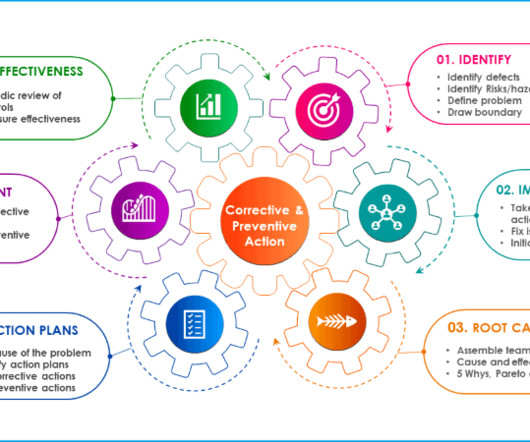
Corrective and preventive actions are identified and implemented as the outcome of investigations such as deviations, product complaints, internal audit observations, risk assessments, stability failure, or other process or system-related non-conformances. The entire unused packaging lot which was quarantined was returned to the supplier.







Let's personalize your content