IPA questions FAERS data interpretation in the Ohio State University study
Express Pharma
MARCH 12, 2025
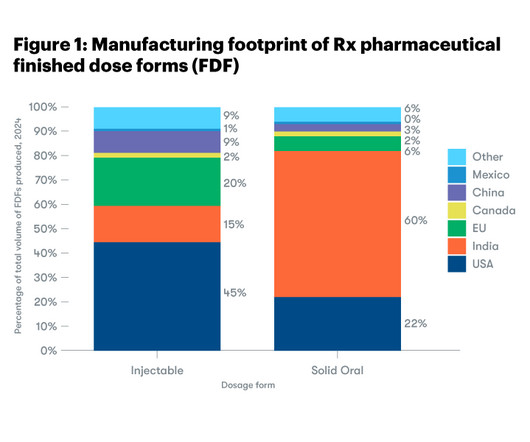
The Indian Pharmaceutical Alliance (IPA) questions the reliance on the US Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) in the Ohio State University report titled, Are All Generic Drugs Created Equal? An Empirical Analysis of Generic Drug Manufacturing Location and Serious Drug Adverse Events.















Let's personalize your content